Drug-Resistant Tuberculosis (DR-TB) remains a major public health challenge in India. Under the National TB Elimination Programme (NTEP), the Programmatic Management of Drug-Resistant TB (PMDT) provides standardized, evidence-based protocols aligned with WHO recommendations and adapted to Indian settings.

•Mono-Resistant TB (MR TB): TB caused by M. TB strains that are resistant to one first- line anti-TB drug only.
•Isoniazid-Resistant TB (Hr-TB): TB is caused by M. TB strains that are resistant to isoniazid, and resistance to rifampicin has been ruled out.
•Poly-Drug Resistant TB (PDR-TB): TB caused by M. TB strains that are resistant to more than one first-line anti-TB drug other than HR and RR.
•Rifampicin Resistant TB (RR-TB): TB caused by M. TB strains that are resistant to R, detected using phenotypic or genotypic methods, with or without resistance to other anti-TB drugs. It includes any resistance to R in the form of mono-resistance, poly-resistance, MDR-TB, or XDR-TB
• Multidrug-Resistant TB (MDR-TB): TB caused by M. TB strains that are resistant to both H and R with or without resistance to other first-line anti-TB drugs. MDR-TB patients also include subsets of Pre-XDR and XDR TB.
•Pre-Extensively Drug-Resistant TB (Pre-XDR-TB): TB caused by M. TB strains that fulfil the definition of MDR/ RR-TB and are also resistant to any fluoroquinolone.
•Extensively Drug-Resistant TB (XDR-TB): TB caused by M. TB strains that fulfil the definition of MDR/RR-TB and are also resistant to any fluoroquinolone (levofloxacin or moxifloxacin) and at least one additional Group A drug (presently to either bedaquiline or linezolid [or both])
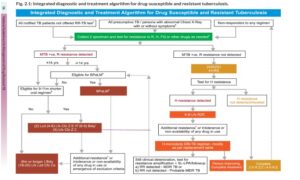
Patient with TruNAAT or CBNAAT M. TB detected and Rif Resistance not detected should be started on HRZE and further samples for culture and LPA should be sent.
Patients with TruNAAT or CBNAAT M. TB detected and Rif Resistance detected should be evaluated for eligibility of BPaLM regimen. If eligible 6 months BPaLM regime should be offered with close ADR monitoring.
Patients with age < 14 years and/or not eligible for BPaLM should be evaluated further for oral shorter or longer regimen.
Once a patient is identified as per the eligibility criteria, the N/DDR-TBC committee will start the patient on BPaLM or 9-11 month shorter oral MDR/RR-TB regimen or 18-20 months longer oral regimen
Figure 1 shows diagnostic and treatment algorithm given by NTEP.
A) BPaLM Regimen
latest NTEP DRTB guidelines says “BPaLM regimen must be the first choice of treatment in eligible patients ≥14 years age with MDR/RR- TB regardless of their FQ resistance status or HIV status.”
The eligibility criteria for BPaLM regimen includes
Inclusion Criteria
•Person with age 14 years & above with new microbiologically confirmed MDR/ RR TB
•H/o of Drug Exposure: Person with exposure of less than one month intake of Bdq, Lzd and/ or Pa in the past or Person with exposure of more than one month intake of Bdq, Lzd and/ or Pa and documented sensitivity to these drugs or Person who had not failed treatment with Bdq or Lzd containing shorter or longer regimen, and sensitivity to these drugs are documented.
•QTcF in ECG is ≤450 ms in males and ≤470 ms in females
Non-lactating women, lactating women but not breast-feeding, non-pregnant women, pregnant women with <20 or <24 weeks gestation and who is willing for medical termination of pregnancy (as per latest MTP gazette notification, as applicable).
Exclusion Criteria & Contra-Indications
•Person with age below 14 years.
•Person with documented resistance to Bdq, Lzd and/ or Pa.
•Person with significant liver dysfunction [LFT (Liver enzymes and/ or total bilirubin); AST/ALT>3.0 x ULN and Total Bilirubin >2.0 x ULN]
•People with severe forms of extrapulmonary-MDR-TB like CNS TB, spinal/ skeletal TB, or disseminated TB (miliary TB or TB with multiorgan involvement)
•Person with significant Cardiac conduction abnormalities in the heart- iincluding in structural heart disease, syncope, long QT syndrome, AV blocks, Reentry arrhythmias etc.
i. Person currently having uncontrolled cardiac arrhythmia that requires medication
ii. Person with history of additional risk factors for Torsade de Pointes.
iii. Baseline QTcF is >450 ms in males & QTcF is >470 ms in females in baseline ECG, and if electrolytes are normal. All MDR/RR-TB patients would be subjected to a thorough pre-treatment evaluation (PTE) –
Clinical Evaluation –
▪ History and physical examination (including previous drug use, alcohol/ substance abuse, family planning methods etc)
▪ Previous history of ATT taken, especially Bdq, Pa, Dlm and Lzd (defined as more than one month exposure)
▪ A thorough clinical examination
▪ Assess nutritional status [Height (m), Weight(kg), BMI]
▪ Neurological evaluation, if required
▪Ophthalmic evaluation, visual acuity, and colour vision test
Laboratory-Based Evaluation –
▪ Random blood sugar (RBS)
▪ HIV testing following counselling
▪ Complete blood count (Hb, TLC, DLC, platelet count)
▪ Liver function tests
▪ Renal function tests
▪ Serum electrolytes (Na, K, Mg, Ca)
▪ Urine pregnancy test (in women of reproductive age group)
▪ Chest X-ray
▪ ECG
BPaLM Dosage
All patients aged 14 years and above would receive the above standard dosage. There is no weight band wise dosing.
1) Bedaquiline Weeks one to two: 400 mg once daily followed by Weeks three to 26/39*: 200 mg three times a week; plus
2) Pretomanid: Weeks one to 26/39: 200 mg daily; plus
3) Linezolid: Weeks one to 26/39: 600 mg once daily; plus
4) Moxifloxacin: Weeks one to 26/39: 400 mg once daily
Pyridoxine (Pdx) to be given as per weight band: 16-29kg 50mg, >30 kg 100mg.
B) 9–11-Month Shorter Oral MDR/RR-TB Regimen
In patients 14 years or above with MDR/RR TB, BPaLM is first preference. The 9–11-month shorter oral MDR/RR-TB regimen is to be preferred over 18-20 months longer M/XDR TB regimen in adults and children with MDR/RR-TB.
Eligibility Criteria-
i. Rifampicin resistance detected.
ii. MDR/RR-TB with FQ resistance not detected.
iii. No history of exposure to previous treatment with second-line medicines in the regimen (Bdq, Lfx, Cfz or Lzd as applicable) for more than one month (unless susceptibility to these medicines is confirmed)
iv. No extensive TB disease
v. No severe forms of extra-pulmonary MDR – TB like CNS TB, spinal/ skeletal TB (miliary TB or TB with multiorgan involvement or disseminated TB)
vi. As Eto has been replaced by Lzd in the regimen therefore the 9–11-month shorter oral MDR/RR- TB regimen with Lzd can be given to pregnant women irrespective of the gestational age with appropriate safety monitoring in consultation with the patient.
vii. For the Lzd containing regimen, thyroid function test is not required in pre-treatment evaluation.
viii. InhA mutation and/or KatG mutation: Lzd containing shorter oral MDR/RR-TB regimen can be given even in case of both KatG & InhA mutations are present. In case of both KatG & InhA mutation, Eto containing shorter oral MDR/RR-TB regimen cannot be given.
ix. Non-lactating women, non-pregnant women, pregnant women with <20 or <24 weeks gestation and who is willing for medical termination of pregnancy (as per latest MTP gazette notification), if Eto is to be used.
The regimen consists of an initial phase of 4 months that may be extended up to 6 months and a continuation phase of 5 months, giving a total duration of 9–11 months. Bdq is used for a duration of 6 months.
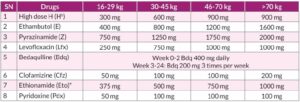
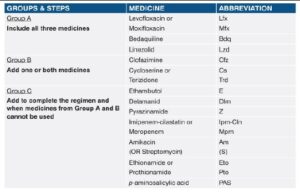
In MDR/RR-TB patients on longer oral M/XDR-TB regimen, all three Group A agents and at least one Group B agent should be included to ensure that treatment starts with at least four TB agents likely to be effective and that at least three agents are included for rest of the treatment if Bdq is stopped. If only one or two Group A agents are used, both Group B agents are to be included. If the regimen cannot be composed with agents from Groups A and B alone, Group C agents are added to complete it as recommended by WHO.
Total duration of longer oral M/XDR-TB regimen is 18–20 months.
Maximum duration of treatment is not more than 20 months. A treatment duration of 15-17 months after culture conversion is suggested for most patients. The duration may be modified according to the patient’s response to treatment.
The dosage of M/XDR-TB drugs for adults used in longer oral M/XDR-TB regimen with replacement drugs from group C in the order of priority customized for India by national experts are enumerated in below given table.
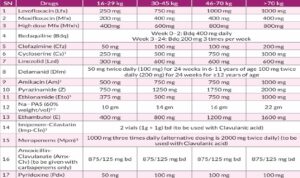
D) Salvage Regimen in XDR-TB
Core Principles
1. Use at least 4–5 likely effective drugs
2. Include all available Group A drugs if susceptible
3. Prefer all-oral regimens
4. Never add a single drug to a failing regimen
5. Consider prior drug exposure as resistance
6. Use phenotypic DST + molecular DST + clinical history
Duration Of Salvage Regimen
•Intensive phase: At least 6–8 months
•Total duration: 18–20 months (may extend to 24 months depending on culture conversion)
•Continue at least 12 months after culture conversion
Carbapenems (Imipenem or Meropenem) are used with clavulanate, administered IV (often twice/thrice daily), indicated in highly resistant XDR cases and reserved for salvage situations due to logistical challenges.
Outcomes in XDR Salvage Therapy
•Treatment success lower than MDR-TB
•Early culture conversion predicts outcome.
Dr. Himanshu Pophale

